Acta medica Lituanica ISSN 1392-0138 eISSN 2029-4174
2021. Online ahead of print DOI: https://doi.org/10.15388/Amed.2021.28.1.7
Age-Related Macular Degeneration: What Do We Know So Far?
Ho Hin Ma*
Lithuanian University of Health Sciences, Medical Academy, Kaunas, Lithuania
Rasa Liutkevičienė
Neuroscience Institute, Lithuanian University of Health Sciences, Medical Academy, Kaunas, Lithuania
Abstract. Ageing is a natural process that everyone experiences and nobody is an exception. With ageing, our body experiences physiological changes. In this article, the focus is made on the physiological changes of our eyes related to ageing and age-related macular degeneration (AMD), which is the most common cause of incurable visual impairment in developed countries. With ageing populations increasing in many countries, more and more patients will have AMD in a foreseeable future. In Eastern Europe, blindness due to AMD, currently, is approximately 20% and there has been an increasing trend depicted in the future. Generally, AMD can be divided into early stages and two forms in an advanced (late) stage. Advanced AMD form includes neovascular AMD (wet) and geographic atrophy (late dry), both of these are associated with substantial, progressive visual impairment. The pathogenesis of AMD is complex and, by far, not completely understood. Multiple factors have been studied, for example: environmental factor, genetic factor (complement factor H), lifestyle. It has been proved that they are linked to higher the risk of developing of AMD, however, the actual pathogenesis is not yet formulated. AMD progression can also be a culprit to certain biochemical events and molecular changes linked to inflammation and pathological angiogenesis. In nowadays, we do have diagnostic methods for both early and late forms of AMD as well as ways to prevent progression of early AMD and wet AMD. However, until now, there is still no treatment for dry AMD. This article is a brief review of AMD and may hopefully lead to some future directions in early diagnostic methods and treating dry AMD.
Key-words: Age-related macular degeneration, drusen, macula lutea, treatment, ageing, genes.
Amžinė geltonosios dėmės degeneracija: ką žinome iki šiol?
Santrauka. Senėjimas yra natūralus visų organizmų procesas ir, deja, nėra išimčių. Senstant mūsų kūnas patiria fiziologinių pokyčių.
Šiame straipsnyje daugiausia dėmesio skiriama fiziologiniams akių pokyčiams, susijusiems su senėjimu, tai yra su amžine geltonosios dėmės degeneracija (AGDD), kuri yra labiausiai paplitusi neišgydomo regėjimo sutrikimo priežastis išsivysčiusiose šalyse. Daugelyje šalių senėjanti populiacija didėja, taigi artimoje ateityje AGDD sirgs vis daugiau pacientų. Rytų Europoje aklumas dėl AGDD dabar yra maždaug 20 proc., o ateityje, manoma, didės.
AGDD formos – pradinė ir vėlyvoji. Vėlyvoji AGDD skirstoma į neovaskulinę AGDD (šlapiąją) ir geografinę atrofiją (sausąją), abi yra susijusios su dideliu progresuojančiu regos aštrumo sutrikimu. AGDD patogenezė yra sudėtinga ir iki šiol nėra iki galo aiški. Manoma, kad jos etiologija daugiaveiksnė ir ją lemia: aplinkos veiksniai, genetiniai veiksniai, pavyzdžiui, komplemento faktorius H, gyvenimo būdas, ir įrodyta jų sąsaja su didesne AGDD išsivystymo rizika. AGDD progresavimas galimas ir dėl tam tikrų biocheminių ir molekulinių pokyčių, susijusių su uždegimu ir patologine angiogeneze. Šiais laikais yra ankstyvosios ir vėlyvosios AGDD formų diagnostikos metodų, taip pat būdų kontroliuoti ankstyvosios ir eksudacinės AGDD progresavimą. Tačiau iki šiol vis dar nėra geografinės AGDD gydymo būdų.
Šiame straipsnyje apžvelgiami pagrindiniai AGDD aspektai ir tikimasi, kad tai paskatins mokslininkus kurti ankstyvus atrofinės AGDD diagnostikos būdus ir gydymo kryptis ateityje.
Raktažodžiai: amžinė geltonosios dėmės degeneracija, drūzos, geltonoji dėmė, gydymas, senėjimas, genai.
* Corresponding author: Ho Hin Ma, Lithuanian University of Health Sciences, Medical Academy, Eiveniu 2, Kaunas, Lithuania, LT-50009, E-mail: mattlscx@gmail.com, hohma0513@kmu.lt, Phone: +370 37 326018
Received: 15/12/2020. Revised: 28/01/2021. Accepted: 28/01/2021
Copyright © 2021 Ho Hin Ma, Rasa Liutkevičienė. Published by Vilnius University Press.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Introduction
It is commonly known that in course of ageing many eye disorders can develop. In our eyes, there is an innermost layer called retina. Macula is a pigmented area located near the centre of the retina, and is responsible for sharp, clear, straight-ahead vision. Despite the peripheral location, macula is part of the central nervous system. As ageing goes, macula experiences structural and blood flow changes that can predispose patients to age-related macular degeneration (AMD). However, one important thing to notice is that advanced age does not necessarily lead to occurrence of AMD.
Every cell in the body experiences changes, for example, the accumulation of lipofuscin within lysosomal compartment of postmitotic cells is a sign of ageing due to iron-catalysed oxidative processes in eyes [1], which prevents phagocytosis of obsolete photoreceptors and results in degeneration of photoreceptors in the periphery of retina [2]. Lipofuscin accumulation does not depend on chronological, rather physiological age [3], in other words, factors affecting our physiology – which implies that diet, genetics, metabolism, lifestyle and habits play a role in age pigment levels in our bodies. Induction by reactive oxygen species (ROS) is considered as one of the contributing factors causing AMD [4,5]. N-Retinylidene-N-Retinylethanolamine (A2E) is a major component found in lipofuscin [6], and the mechanisms behind how A2E leads to RPE cells damage and macular degeneration have lately been proposed [7,8].
In general, there are two types of AMD, one is called dry AMD, while another one is called wet AMD. Compared with wet AMD, the deteriorating process of dry AMD is slower and visual acuity is better preserved [9]. Nevertheless, 20% of legal blindness over the population of AMD belongs to dry AMD [10], which is considerably significant. Atrophic age related macular degeneration (atrophic AMD), also known as dry AMD, makes up approximately 90% of individuals with conditions of macular degeneration [11]. It is better to include this type of classification: early AMD is defined as the presence of drusen and retinal pigmentary abnormalities; late AMD includes dry AMD (geographic atrophy of the retinal pigmentary epithelium in the absence of neovascular AMD) or neovascular AMD (detachment of the retinal pigment epithelium, hemorrhages, and/or scars). In most cases AMD starts as the dry type and in 10–20% of individuals, it progresses to the wet type [12]. There are various and numerous studies about age-related macular degeneration; however, much of these existing studies and statistics do not indicate whether the results are obtained for ‘wet’ AMD or ‘dry’ AMD specifically. Dry AMD also known as nonneovascular AMD or nonexudative AMD, because there is an absence of abnormal blood vessels growing underneath the retina, leading to leakage of blood or serum. Rather, dry AMD is associated with degeneration of retinal pigment epithelium (RPE) cells and photoreceptor cells, and diagnosed and characterised by the formation and growth of small yellow deposits known as drusen, which dysfunction and damage the Retinal Pigment Epithelium cells, continuation of rod loss and followed by degeneration of cones and eventually only cones of the fovea remain and die off [13]. Moreover, leakage of fluid beneath RPE and Bruch membrane may occur [14].
Upon reaching the phase of losing of choriocapillaris, RPE, and photoreceptors, the stage is known as geographic atrophy, leading to irreversible loss of vision. Geographic Atrophy is an advanced form of dry AMD and characterised by the presence of sharply demarcated atrophic outer retina. To date, although there are no approved medical treatments for both dry AMD and geographic atrophy, some are undergoing clinical trials [15]. Dry AMD can not only develop into geographic atrophy, but 4–12% of cases per year of dry AMD also can develop to choroidal neovascularization (CNV), a characteristic of wet AMD [16], due to the cracking of more fragmented and calcified Bruch’s membrane [17].
In this article, we review the predisposed risk factors of AMD and its etiopathogenesis in order to deduce possibilities of early diagnosis, treatment, and prevention of both early and advanced age-related macular degeneration.
Composition of drusen
Drusen are composed of lipids, RPE cells fragments, lipofuscin, dendritic cells, potent antigen-presenting cells [18]. In addition, hydroxyapatite (HAP) spherules [19], HAP nodules, trace elements like Zinc and Copper are also found [20]. All of these can be a reason how drusen form and increase in size. Nevertheless, interestingly, they are made up of various proteins, for example: glycoproteins [21], chaperone proteins, Apolipoprotein E, vitronectin, amyloid P, C5, and C5b09 complement complex. Taken together, the information shows that drusen share some features of proteins misfolding in other neurodegenerative conditions [22].
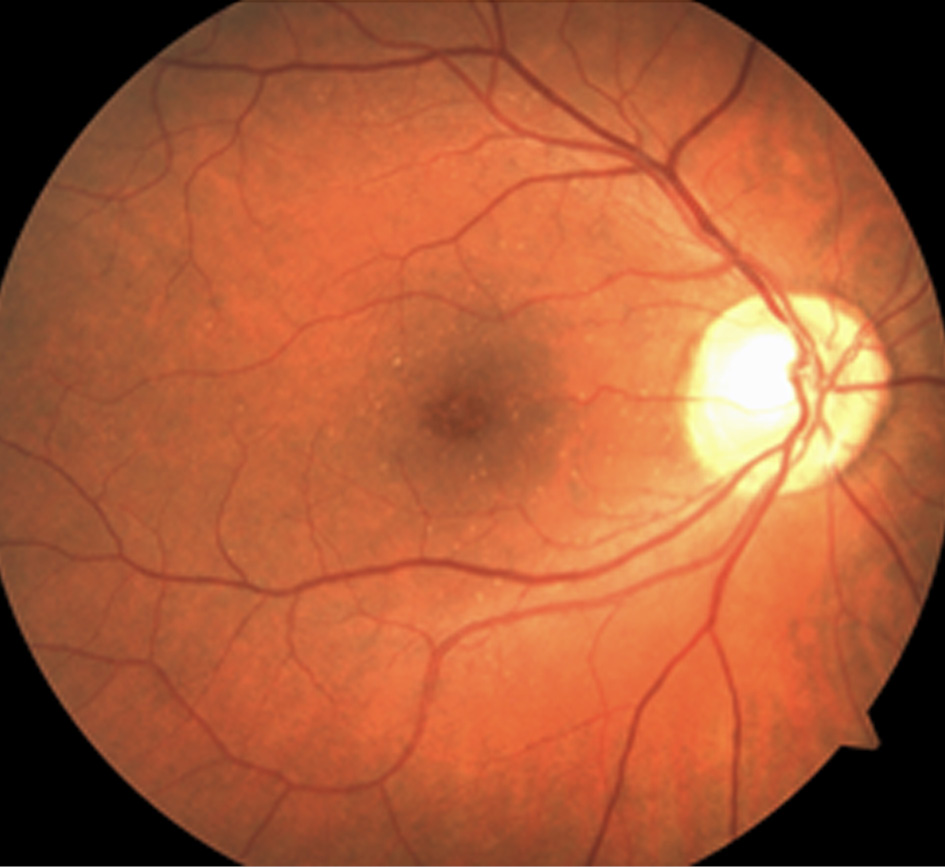
Drusen can be categorized into six types and they are as follows: hard (nodular) drusen, soft (exudative) drusen, basal linear deposits (diffuse drusen or BLinD), cuticular drusen (previous term: basal laminar drusen or BLamD), and reticular pseudodrusen (reticular drusen, subretinal drusenoid deposits). Universally, diffuse drusen and early cuticular drusen are defined as thresholds of the onset of early age-related macular degeneration [23]. Hard drusen (Fig.1) are small, yellow nodules and there is a possibility for them to progress to dry AMD [24]. They are observed in both normal ageing eyes and AMD patients [25], while soft drusen appear as larger with indistinguishable borders, which can lead to certain parts of RPE being separated [24].
Figure 1. AMD (hard drusen in the fovea) in the right eye
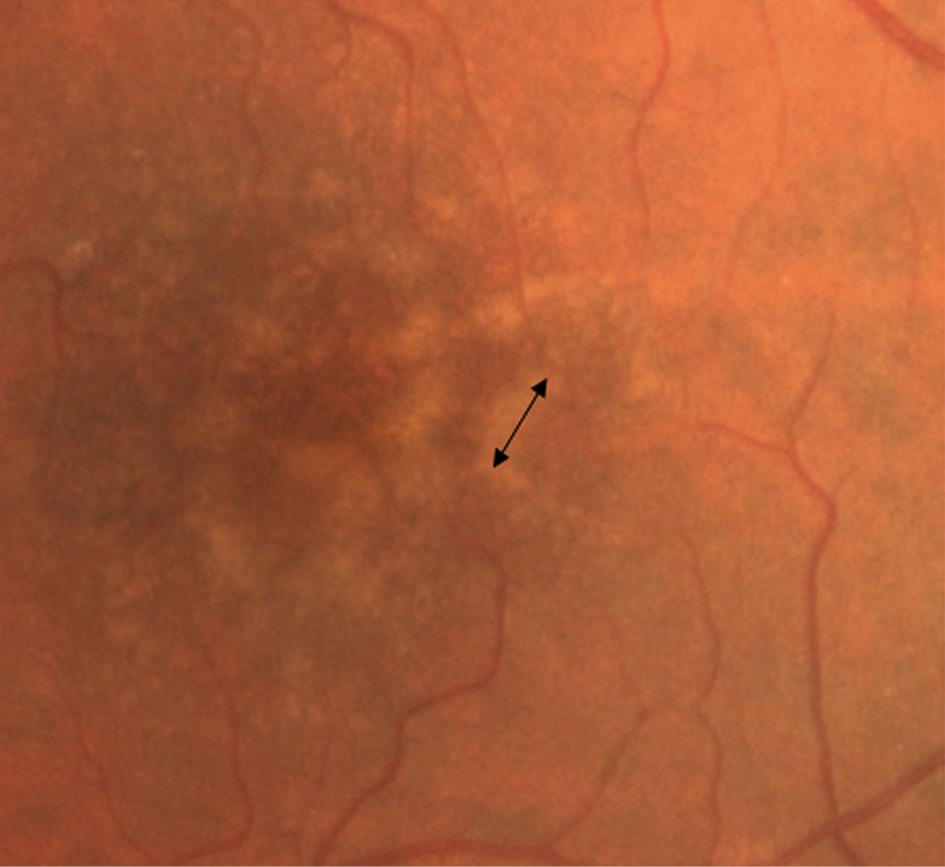
Figure 2. AMD (soft drusen in the fovea with shown diameter of the drusen)
Drusen themselves do not cause any forms of AMD, but their presence in time course definitely increase the risk of developing of AMD. Human drusen consist of lipids primarily derived from photoreceptor cells and serum, and proteins apparently primarily derived from choroidal cells and serum [26]. To date, it is not known how drusen occur, their pathogenesis and ability to vary in location still remain a mystery. Some proposed that formation of drusen can be traced back to the fact that phagocytosis of RPE declines with age [27], and it’s believed that drusen is created by lipofuscin from the oxidative stress accumulated between Bruch’s membrane and (diseased) RPE [28,29].
Macula lutea
Macula lutea, in homo sapiens, has a diameter approximately 5.5 mm and is subdivided into umbo, foveola, foveal avascular zone, fovea, parafovea, and perifovea regions. It is located at the posterior pole of the eye on the retina and is responsible for central vision. Photoreceptor density is above average throughout macula, therefore when experiencing AMD, central vision is usually faded out or lost. Macula houses three pigments including lutein (L), zeaxanthin (Z) and meso-zeaxanthin (MZ). All of these carotenoids can help in vision and possibly protection from AMD due to their antioxidant properties [30]. Macular carotenoids can be obtained via diet, in other words, the risk of developing of advanced AMD can be reduced by having a diet with macular carotenoids [31], for example, in green leafy vegetables, eggs, corn, red peppers, and some other red, yellow, orange, and dark green fruits and vegetables [31]. Macular carotenoids cannot be synthesized de novo, therefore have to be obtained through diet. Potentially, low levels of serum carotenoids can be a signal alerting someone might be associated with a higher risk of having wet AMD and meanwhile, serum carotenoids can be used as a biomarker of early onset of both dry and wet AMD [32]. Interestingly, there is a positive correlation between levels of carotenoids and neuro-cognitive functions [33], and neuro-cognitive function is less affected when retinal carotenoids are higher even though ageing proceeds. Benefits of retinal carotenoids are not only limited to slow down the decline of neuro-cognitive functions, but also decline the probability of AMD development and advancement [34,35,36] and demonstrate anti-oxidative and anti-inflammatory properties [37,38]. Besides obtaining retinal carotenoids through diet, dietary polyphenols (DPs) have also been reported to be advantageous for our vision [39].
Macula houses cone cells, which are responsible for colour vision under bright light conditions. In dry AMD, cones appear more likely to survive compared to rods [40], and like in other retinal diseases, rod cells’ apoptosis precedes cone cells’ death. It has been found that abnormalities in distal cone axons could be an indicator to early AMD [41]. AMD is a multifactorial disease of ageing. Many researchers have already proposed the pathogenesis of AMD, and it is summarized as follows: lipofuscin accumulation and retinal epithelial cell damage [42], oxidative stress [43], lipids and lipid peroxidation products [44], chronic inflammation [45], abnormal extracellular matrix [46], and metabolic distress [47]. The risk factors for AMD include: oxidative stress [48], age-related changes in ocular hemodynamics [9], individuals with myocardial infarction [84], genetic predisposition [49], gender [50], ethnicity [51,52,53], colour of iris [54], prolonged exposure of sunlight [55], smoking [56], hypertension [57,58], eating habits [59,60], serum antioxidants and those in diet [61], increased inflammatory markers in blood [62], presence of AMD in the other eye [63], lack of exercise [64], vigorous exercise [65], and accumulation of lipofuscin [66]. Psychological stress is potentially a risk factor for AMD [67]. To date, there is no increased risk of progression to dry AMD after post-cataract surgery [68,69]. Some research showed that blue light damages retina because of higher photon energy [70,71], and it’s true; however, the blue light from electronic devices is dim and therefore, exposure of blue light from electronic devices is not considered as a risk to advance the AMD development. As an old saying goes, “prevention is better than cure”, researchers show that weight loss can prevent the development of AMD [72]; however, it still takes time to see whether there is a reverse relationship, in other words, whether weight gain will increase the risk of AMD is not yet known and needs to be investigated [73].
Generally, it is agreed that the appearance of drusen deposits at the base of the RPE is the sign of early AMD. Studies [19,20] have found that there is a linkage between calcium and early onset of AMD and the formation of drusen; however, there is inconsistency in their results [74,75]; whether calcium intake can raise or lower the risk, it’s yet to be concluded. To date, studies have been reported that mice are not good animal models to study dry AMD because drusen are rarely seen in mice owing to the simple structure of their basement membrane and lipofuscin production process is different compared with that in humans. For animal models, it’s been reported that Macaca fascicularis [76] and Macaca mulatta [77] are beneficial in studying the developmental process and formation of drusen in dry AMD. Notably, there is no reported advancement of AMD observed in non homo sapiens primates [78]. Perhaps, by studying the genomics of the animal models mentioned before, certain roles of genes could be uncovered to halt the progression of dry AMD or even its onset. Dry AMD in no doubt affects the quality of life negatively, for example, a person with dry AMD will experience difficulties in reading, driving, and recognizing faces. Currently, there are no treatments for dry AMD and geographic atrophy, an advancement form of dry AMD; however, there are some clinical trials of dry AMD ongoing [79,80,81] and proposed, all of these sound promising and hopeful. Age-related macular degeneration, as mentioned before, is a multifactorial disease and genetic factors play a role in its development and advancement. 36 genes were discovered and identified to be involved in the risk factors and pathogenesis of AMD [82]. Among them, complement factor H (CFH) was the most significant gene found to be related to AMD [83] and is always brought up in other studies in developing lipoprotein treatment for AMD patients [84], and not limited to understanding the pathology of early AMD [85]. Not long ago, studies discovered and proved that there are another 2 gene candidates, PRMT6 and LSS, to study the functional aspects of geographic atrophy [86]. Moreover, recently in the UK, a genetic approach was adopted in the field of atrophic AMD targeting complement factors (NCT03846193) and so far no results are yet available. With the recent advances in bioengineering [87,88] and stem cells technology [27,89,90,91,92], individuals with exudative AMD are reported to have improvement in their visions. The mechanisms of nonexudative AMD are complex; regardless of challenges mentioned in the study, certain ways with the usage of stem cell derived RPE transplant seems to be a promising solution [92].
Possible diagnosis and treatment for early AMD and atrophic AMD
To date, there have been several treatments of macular degeneration put into practice (Subretinal Transplantation of Embryonic Stem Cell–Derived Retinal Pigment Epithelium for the Treatment of Macular Degeneration; Retinal pigment epithelium transplantation: concepts, challenges, and future prospects; Phase I/IIa Clinical Trial of Human Embryonic Stem Cell (hESC)-Derived Retinal Pigmented Epithelium (RPE, OpRegen) Transplantation in Advanced Dry Form Age-Related Macular Degeneration (AMD): Interim Results), and more genetic polymorphisms (association of genetic variants at CETP, AGER, and CYP4F2 locus with the risk of atrophic age‐related macular degeneration) have been discovered lately to reveal the interactions of the genes to the AMD pathogenesis.
Currently, there are medications/treatment methods for wet AMD, and it’s definitely possible to identify SNP of wet AMD and most ideally, several biomarkers can be deduced and revealed. Since at the advanced stage AMD develops into either dry AMD or wet AMD, by comparing the SNPs in the patients with those in the healthy group, which have been sorted, possibly and hopefully the SNP of dry AMD and the mystery of ageing can be deduced, with the overlap and elimination of some SNPs. In the past, in patients with AMD the connection has been shown with hypertension, dyslipidemia, as well as diabetes mellitus; all of these can be traced to a common factor - blood type, should be evaluated, as well.
Discussion
Ageing is not a pathology, but a process that every organism is experiencing. Prevention is better than cure, AMD occurs less often in people who exercise on a regular basis, avoid smoking, and eat nutritious foods including green leafy vegetables and fish. There is an importance to further investigate the processes of normal ageing in the human retina and the changing in the eyes of individuals with AMD. It’s also important to study the correlation between trace elements and AMD regardless inconsistencies mentioned before on the role of trace elements in ageing and AMD progression. For example, in AMD patients, both retinal zinc and iron levels decrease in RPE, hence potentially both of these can be an indicator to invite individuals to have a comprehensive eye exam. Further, confirmation if BLam or Blin deposits (basal laminar deposit, BLamD; basal linear deposit, BLinD) between the cell membrane and the basement membrane of the RPE appear in individuals is also a way, because these deposits precede the emergence of drusen [93]. Several years ago, research studies demonstrated that projected AMD population by 2020 at an increasing trend (94,95,96), and more recently, on the other hand, studies conducted in the USA [97] and Europe discovered that there is a decreasing prevalence of AMD in regions and continents [98]. By combining the data, one can observe inconsistency in the prevalence of AMD. It is highly suggested and recommended to run a meta-analysis to follow up and unmask and reflect the real phenomenon of AMD in regions and continents nowadays, also to see whether nowadays the prevalence of AMD is declined or actually increased as expected, and whether there is a trend of individuals of developing AMD at a later age or an earlier age. It’s worthy of note that the “A” in AMD refers to the biological age instead of the chronological age. In other words, a healthy lifestyle, a healthy dietary habits, good mental health, stress-free environments [99, 100], not just genetic factors, play a role in slowing down ageing and ageing associated diseases. It is recommended to conduct an investigation whether there is a discrepancy between the biological age and the chronological age in individuals with different stages of AMD, and whether protecting telomeres from damage can treat AMD [8]. An interesting thing is that for 10–20% of patients with dry AMD it develops into wet AMD – how does it develop? What genes play certain roles to make it happen? Can the conversion of dry AMD to wet AMD be a cure for dry AMD? Investigations are recommended to discover the possibilities, forward the discoveries, and unfold the mystery.
Conclusion
Regardless of various risk factors and candidate genes have been discovered and mentioned above, as well as associations drawn to atrophic AMD, it still makes up the majority cases of age-related macular degeneration. Yet, a broader understanding of how interactions of factors, the causative factors of dry AMD, and correlation between genetics and pathological condition remains a mystery. Ageing is a complicated process with multiple factors, let alone age-related macular degeneration. By far, the pathogenesis, causation and correlation of both environmental and genetic factors of atrophic AMD are awaiting to be discovered and reviewed. Hopefully in the future, a feasible cure and treatment can be provided.
Proprietary interests or conflicts: none of the authors has any proprietary interests or conflicts of interest related to this submission.
This submission has not been published anywhere previously, and it is not simultaneously being considered for any other publication.
References
- Dolman CL, MacLeod PM. Lipofuscin and its relation to aging. In Advances in cellular neurobiology 1981 Jan 1 (Vol. 2, pp. 205-247). Elsevier. DOI: https://doi.org/10.1016/B978-0-12-008302-2.50011-0
- Nilsson SE, Sundelin SP, Wihlmark U, Brunk UT. Aging of cultured retinal pigment epithelial cells: oxidative reactions, lipofuscin formation and blue light damage. Documenta Ophthalmologica. 2003 Jan;106(1):13-6.DOI: https://doi.org/10.1023/A:1022419606629.
- Gerstbrein B, Stamatas G, Kollias N, Driscoll M. In vivo spectrofluorimetry reveals endogenous biomarkers that report healthspan and dietary restriction in Caenorhabditis elegans. Aging cell. 2005 Jun;4(3):127-37. DOI: https://doi.org/10.1111/j.1474-9726.2005.00153.x
- Dunaief JL. Iron induced oxidative damage as a potential factor in age-related macular degeneration: the Cogan Lecture. Investigative ophthalmology & visual science. 2006 Nov 1;47(11):4660-4. DOI: https://doi.org/10.1167/iovs.06-0568
- Beatty S, Koh HH, Phil M, Henson D, Boulton M. The role of oxidative stress in the pathogenesis of age-related macular degeneration. Survey of ophthalmology. 2000 Sep 1;45(2):115-34. DOI: https://doi.org/10.1016/s0039-6257(00)00140-5
- Sparrow JR, Fishkin N, Zhou J, Cai B, Jang YP, Krane S, Itagaki Y, Nakanishi K. A2E, a byproduct of the visual cycle. Vision research. 2003 Dec 1;43(28):2983-90. DOI: https://doi.org/10.1016/s0042-6989(03)00475-9
- Parmar VM, Parmar T, Arai E, Perusek L, Maeda A. A2E-associated cell death and inflammation in retinal pigmented epithelial cells from human induced pluripotent stem cells. Stem cell research. 2018 Mar 1;27:95-104. DOI: https://doi.org/10.1016/j.scr.2018.01.014
- Wang J, Feng Y, Han P, Wang F, Luo X, Liang J, Sun X, Ye J, Lu Y, Sun X. Photosensitization of A2E triggers telomere dysfunction and accelerates retinal pigment epithelium senescence. Cell death & disease. 2018 Feb 7;9(2):1-4. DOI: https://doi.org/10.1038/s41419-017-0200-7
- Ehrlich R, Harris A, Kheradiya NS, Winston DM, Ciulla TA, Wirostko B. Age-related macular degeneration and the aging eye. Clinical interventions in aging. 2008 Sep;3(3):473. DOI: https://doi.org/10.2147/cia.s2777
- Michalska-Małecka K, Kabiesz A, Nowak M, Śpiewak DJ. Age related macular degeneration–challenge for future: pathogenesis and new perspectives for the treatment. European Geriatric Medicine. 2015 Feb 1;6(1):69-75. DOI: https://doi.org/10.1016/j.eurger.2014.09.007
- Cheng WS, Lu DW, Chiang CH, Chang CJ. Overview of clinical trials for dry age-related macular degeneration. Journal of Medical Sciences. 2017 Jul 1;37(4):121. DOI: https://doi.org/10.4103/jmedsci.jmedsci_115_16
- Mehta JL, Dhalla NS, editors. Biochemical basis and therapeutic implications of angiogenesis. London, New York: Springer; 2013 Mar 29. DOI: https://doi.org/10.1007/978-3-319-61115-0
- Curcio CA, Medeiros NE, Millican CL. Photoreceptor loss in age-related macular degeneration. Investigative ophthalmology & visual science. 1996 Jun 1;37(7):1236-49.
- Kolb H. Simple anatomy of the retina by helga kolb. Webvision: The Organization of the Retina and Visual System. 2011 Oct 8.
- Nebbioso M, Lambiase A, Cerini A, Limoli PG, La Cava M, Greco A. Therapeutic approaches with intravitreal injections in geographic atrophy secondary to age-related macular degeneration: current drugs and potential molecules. International journal of molecular sciences. 2019 Jan;20(7):1693. DOI: https://doi.org/10.3390/ijms20071693
- Hayat MA, editor. Autophagy: Cancer, Other Pathologies, Inflammation, Immunity, Infection, and Aging: Volume 12. Academic Press; 2017 Sep 7. DOI: https://doi.org/10.1016/C2014-0-03338-2
- Spraul CW, Grossniklaus HE. Characteristics of Drusen and Bruch’s membrane in postmortem eyes with age-related macular degeneration. Archives of ophthalmology. 1997 Feb 1;115(2):267-73. DOI: https://doi.org/10.1001/archopht.1997.01100150269022
- Hageman GS, Luthert PJ, Chong NV, Johnson LV, Anderson DH, Mullins RF. An integrated hypothesis that considers drusen as biomarkers of immune-mediated processes at the RPE-Bruch’s membrane interface in aging and age-related macular degeneration. Progress in retinal and eye research. 2001 Nov 1;20(6):705-32. DOI: https://doi.org/10.1016/s1350-9462(01)00010-6
- Thompson RB, Reffatto V, Bundy JG, Kortvely E, Flinn JM, Lanzirotti A, Jones EA, McPhail DS, Fearn S, Boldt K, Ueffing M. Identification of hydroxyapatite spherules provides new insight into subretinal pigment epithelial deposit formation in the aging eye. Proceedings of the National Academy of Sciences. 2015 Feb 3;112(5):1565-70. DOI: https://doi.org/10.1073/pnas.1413347112
- Pilgrim MG, Lengyel I, Lanzirotti A, Newville M, Fearn S, Emri E, Knowles JC, Messinger JD, Read RW, Guidry C, Curcio CA. Subretinal pigment epithelial deposition of drusen components including hydroxyapatite in a primary cell culture model. Investigative ophthalmology & visual science. 2017 Feb 1;58(2):708-19. DOI: https://doi.org/10.1167/iovs.16-21060
- D’souza Y, Jones CJ, Bonshek R. Glycoproteins of drusen and drusen-like lesions. Journal of molecular histology. 2008 Feb 1;39(1):77-86. DOI: https://doi.org/10.1007/s10735-007-9130-5
- Sauer T, Patel M, Chan CC, Tuo J. Unfolding the therapeutic potential of chemical chaperones for age-related macular degeneration. Expert review of ophthalmology. 2008 Feb 1;3(1):29-42. DOI: https://doi.org/10.1586/17469899.3.1.29
- Sarks S, Cherepanoff S, Killingsworth M, Sarks J. Relationship of basal laminar deposit and membranous debris to the clinical presentation of early age-related macular degeneration. Investigative ophthalmology & visual science. 2007 Mar 1;48(3):968-77. DOI: https://doi.org/10.1167/iovs.06-0443
- Abdelsalam A, Del Priore L, Zarbin MA. Drusen in age-related macular degeneration: pathogenesis, natural course, and laser photocoagulation–induced regression. Survey of ophthalmology. 1999 Jul 1;44(1):1-29. DOI: https://doi.org/10.1016/s0039-6257(99)00072-7.
- Buch H, Nielsen NV, Vinding T, Jensen GB, Prause JU, la Cour M. 14-year incidence, progression, and visual morbidity of age-related maculopathy: the Copenhagen City Eye Study. Ophthalmology. 2005 May 1;112(5):787-98. DOI: https://doi.org/10.1016/j.ophtha.2004.11.040
- Booij JC, Baas DC, Beisekeeva J, Gorgels TG, Bergen AA. The dynamic nature of Bruch’s membrane. Progress in retinal and eye research. 2010 Jan 1;29(1):1-8. DOI: https://doi.org/10.1016/j.preteyeres.2009.08.003
- Inana G, Murat C, An W, Yao X, Harris IR, Cao J. RPE phagocytic function declines in age-related macular degeneration and is rescued by human umbilical tissue derived cells. Journal of translational medicine. 2018 Dec;16(1):1-5. DOI: https://doi.org/10.1186/s12967-018-1434-6
- Sohal RS, Brunk UT. Lipofuscin as an indicator of oxidative stress and aging. Advances in experimental medicine and biology. 1989 Jan 1;266:17-26. DOI: https://doi.org/10.1007/978-1-4899-5339-1_2
- Algvere PV, Kvanta A, Seregard S. Drusen maculopathy: a risk factor for visual deterioration. Acta ophthalmologica. 2016 Aug;94(5):427-33. DOI: https://doi.org/10.1111/aos.13011
- Nolan JM, Meagher K, Kashani S, Beatty S. What is meso-zeaxanthin, and where does it come from?. Eye. 2013 Aug;27(8):899-905. DOI: https://doi.org/10.1038/eye.2013.98
- Wise J. Diet rich in carotenoids is linked to reduced risk of advanced age related macular degeneration. Bmj. 2015 Oct 9;351. DOI: https://doi.org/10.1136/bmj.h5384
- Zhou H, Zhao X, Johnson EJ, Lim A, Sun E, Yu J, Zhang Y, Liu X, Snellingen T, Shang F, Liu N. Serum carotenoids and risk of age-related macular degeneration in a Chinese population sample. Investigative ophthalmology & visual science. 2011 Jun 1;52(7):4338-44. DOI: https://doi.org/10.1167/iovs.10-6519
- Walk AM, Edwards CG, Baumgartner NW, Chojnacki MR, Covello AR, Reeser GE, Hammond BR, Renzi-Hammond LM, Khan NA. The role of retinal carotenoids and age on neuroelectric indices of attentional control among early to middle-aged adults. Frontiers in aging neuroscience. 2017 Jun 9;9:183. DOI: https://doi.org/10.3389/fnagi.2017.00183
- Wu J, Cho E, Willett WC, Sastry SM, Schaumberg DA. Intakes of lutein, zeaxanthin, and other carotenoids and age-related macular degeneration during 2 decades of prospective follow-up. JAMA ophthalmology. 2015 Dec 1;133(12):1415-24. DOI: https://doi.org/10.1001/jamaophthalmol.2015.3590
- Bungau S, Abdel-Daim MM, Tit DM, Ghanem E, Sato S, Maruyama-Inoue M, Yamane S, Kadonosono K. Health benefits of polyphenols and carotenoids in age-related eye diseases. Oxidative medicine and cellular longevity. 2019 Feb 12;2019. DOI: https://doi.org/10.1155/2019/9783429
- Cho KS, Shin M, Kim S, Lee SB. Recent advances in studies on the therapeutic potential of dietary carotenoids in neurodegenerative diseases. Oxidative medicine and cellular longevity. 2018 Oct;2018. DOI: https://doi.org/10.1155/2018/4120458
- Parmeggiani F, Romano MR, Costagliola C, Semeraro F, Incorvaia C, D’Angelo S, Perri P, De Palma P, De Nadai K, Sebastiani A. Mechanism of inflammation in age-related macular degeneration. Mediators of inflammation. 2012 Oct;2012. DOI: https://doi.org/10.1155/2012/546786
- Shahandeh A, Purushothuman S, Martin K, Graham M, Johnstone DM, Milward EA. Anti-Oxidant Phytochemicals As Potential Treatments For Age-Related Macular Degeneration. Journal of antioxidant activity. 2015 Jun 10;1(1):29. DOI: https://doi.org/10.14302/issn.2471-2140.jaa-14-616
- Pawlowska E, Szczepanska J, Koskela A, Kaarniranta K, Blasiak J. Dietary polyphenols in age-related macular degeneration: Protection against oxidative stress and beyond. Oxidative medicine and cellular longevity. 2019 Mar 24;2019. DOI: https://doi.org/10.1155/2019/9682318
- Holz FG, Gross-Jendroska M, Eckstein A, Hogg CR, Arden GB, Bird AC. Colour contrast sensitivity in patients with age-related Bruch’s membrane changes. German journal of ophthalmology. 1995 Nov 1;4(6):336-41.
- Shelley EJ, Madigan MC, Natoli R, Penfold PL, Provis JM. Cone degeneration in aging and age-related macular degeneration. Archives of ophthalmology. 2009 Apr 1;127(4):483-92. DOI: https://doi.org/10.1001/archophthalmol.2008.622
- Brunk UT, Terman A. Lipofuscin: mechanisms of age-related accumulation and influence on cell function. Free Radical Biology and Medicine. 2002 Sep 1;33(5):611-9. DOI: https://doi.org/10.1016/s0891-5849(02)00959-0
- Jarrett SG, Boulton ME. Consequences of oxidative stress in age-related macular degeneration. Molecular aspects of medicine. 2012 Aug 1;33(4):399-417. DOI: https://doi.org/10.1016/j.mam.2012.03.009
- Kopitz J, Holz FG, Kaemmerer E, Schutt F. Lipids and lipid peroxidation products in the pathogenesis of age-related macular degeneration. Biochimie. 2004 Nov 1;86(11):825-31. DOI: https://doi.org/10.1016/j.biochi.2004.09.029
- Chen M, Xu H. Parainflammation, chronic inflammation, and age‐related macular degeneration. Journal of leukocyte biology. 2015 Nov;98(5):713-25. DOI: https://doi.org/10.1189/jlb.3RI0615-239R
- Fernandez-Godino R, Bujakowska KM, Pierce EA. Changes in extracellular matrix cause RPE cells to make basal deposits and activate the alternative complement pathway. Human molecular genetics. 2018 Jan 1;27(1):147-59. DOI: https://doi.org/10.1093/hmg/ddx392
- Brown CN, Green BD, Thompson RB, Den Hollander AI, Lengyel I. Metabolomics and age-related macular degeneration. Metabolites. 2019 Jan;9(1):4. DOI: https://doi.org/10.3390/metabo9010004
- Friedman E. A hemodynamic model of the pathogenesis of age-related macular degeneration. American journal of ophthalmology. 1997 Nov 1;124(5):677-82. DOI: https://doi.org/10.1016/s0002-9394(14)70906-7
- DeAngelis MM, Owen LA, Morrison MA, Morgan DJ, Li M, Shakoor A, Vitale A, Iyengar S, Stambolian D, Kim IK, Farrer LA. Genetics of age-related macular degeneration (AMD). Human molecular genetics. 2017 Aug 1;26(R1):R45-50. DOI: https://doi.org/10.1093/hmg/ddx228
- Sasaki M, Harada S, Kawasaki Y, Watanabe M, Ito H, Tanaka H, Takeuchi A, Tsubota K, Takebayashi T, Nishiwaki Y, Kawasaki R. Gender-specific association of early age-related macular degeneration with systemic and genetic factors in a Japanese population. Scientific reports. 2018 Jan 15;8(1):1-8. DOI: https://doi.org/10.1038/s41598-017-18487-4
- Klein R, Klein BE, Knudtson MD, Wong TY, Cotch MF, Liu K, Burke G, Saad MF, Jacobs Jr DR. Prevalence of age-related macular degeneration in 4 racial/ethnic groups in the multi-ethnic study of atherosclerosis. Ophthalmology. 2006 Mar 1;113(3):373-80. DOI: https://doi.org/10.1016/j.ophtha.2005.12.013
- Vanderbeek BL, Zacks DN, Talwar N, Nan B, Musch DC, Stein JD. Racial differences in age-related macular degeneration rates in the United States: a longitudinal analysis of a managed care network. American journal of ophthalmology. 2011 Aug 1;152(2):273-82. DOI: https://doi.org/10.1016/j.ajo.2011.02.004
- Klein R. Race/ethnicity and age-related macular degeneration. American journal of ophthalmology. 2011 Aug 1;152(2):153-4. DOI: https://doi.org/10.1016/j.ajo.2011.02.016.
- Frank RN, Puklin JE, Stock C, Canter LA. Race, iris color, and age-related macular degeneration. Transactions of the American Ophthalmological Society. 2000;98:109.
- Sui GY, Liu GC, Liu GY, Gao YY, Deng Y, Wang WY, Tong SH, Wang L. Is sunlight exposure a risk factor for age-related macular degeneration? A systematic review and meta-analysis. British Journal of Ophthalmology. 2013 Apr 1;97(4):389-94. DOI: https://doi.org/10.1136/bjophthalmol-2012-302281
- Velilla S, García-Medina JJ, García-Layana A, Dolz-Marco R, Pons-Vázquez S, Pinazo-Durán MD, Gómez-Ulla F, Arévalo JF, Díaz-Llopis M, Gallego-Pinazo R. Smoking and age-related macular degeneration: review and update. Journal of ophthalmology. 2013 Dec 4;2013. DOI: https://doi.org/10.1155/2013/895147
- Katsi VK, Marketou ME, Vrachatis DA, Manolis AJ, Nihoyannopoulos P, Tousoulis D, Vardas PE, Kallikazaros I. Essential hypertension in the pathogenesis of age-related macular degeneration: a review of the current evidence. Journal of hypertension. 2015 Dec 1;33(12):2382-8. DOI: https://doi.org/10.1097/HJH.0000000000000766
- Cougnard-Grégoire A, Delyfer MN, Korobelnik JF, Rougier MB, Malet F, Le Goff M, Dartigues JF, Colin J, Barberger-Gateau P, Delcourt C. Long-term blood pressure and age-related macular degeneration: the ALIENOR study. Investigative ophthalmology & visual science. 2013 Mar 1;54(3):1905-12. DOI: https://doi.org/10.1167/iovs.12-10192.
- Chiu CJ, Chang ML, Zhang FF, Li T, Gensler G, Schleicher M, Taylor A. The relationship of major American dietary patterns to age-related macular degeneration. American journal of ophthalmology. 2014 Jul 1;158(1):118-27. DOI: https://doi.org/10.1016/j.ajo.2014.04.016
- Hogg RE, Woodside JV. Mediterranean Diet and Age-Related Macular Degeneration: Is It Time to Attempt Dietary Modification?. Ophthalmology. 2019 Mar 1;126(3):391-2. DOI: https://doi.org/10.1016/j.ophtha.2018.09.032.
- Mares-Perlman JA, Brady WE, Klein R, Klein BE, Bowen P, Stacewicz-Sapuntzakis M, Palta M. Serum antioxidants and age-related macular degeneration in a population-based case-control study. Archives of ophthalmology. 1995 Dec 1;113(12):1518-23. DOI: https://doi.org/10.1001/archopht.1995.01100120048007
- Colak E, Majkic-Singh N, Zoric L, Radosavljevic A, Kosanovic-Jakovic N. The role of CRP and inflammation in the pathogenesis of age-related macular degeneration. Biochemia medica. 2012 Feb 15;22(1):39-48. DOI: https://doi.org/10.11613/bm.2012.005
- Cook HL, Patel PJ, Tufail A. Age-related macular degeneration: diagnosis and management. British medical bulletin. 2008 Mar 1;85(1):127-49. DOI: https://doi.org/10.1093/bmb/ldn012
- McGuinness MB, Le J, Mitchell P, Gopinath B, Cerin E, Saksens NT, Schick T, Hoyng CB, Guymer RH, Finger RP. Physical activity and age-related macular degeneration: a systematic literature review and meta-analysis. American journal of ophthalmology. 2017 Aug 1;180:29-38. DOI: https://doi.org/10.1016/j.ajo.2017.05.016
- Rim TH, Kim HK, Kim JW, Lee JS, Kim DW, Kim SS. A Nationwide cohort study on the association between past physical activity and neovascular age-related macular degeneration in an East Asian population. JAMA ophthalmology. 2018 Feb 1;136(2):132-9. DOI: https://doi.org/10.1001/jamaophthalmol.2017.5682.
- Wolf G. Lipofuscin and macular degeneration. Nutrition reviews. 2003 Oct 1;61(10):342-6. DOI: https://doi.org/10.1301/nr.2003.oct.342-346
- Dougherty BE, Cooley SS, Davidorf FH. Measurement of perceived stress in age related macular degeneration. Optometry and vision science: official publication of the American Academy of Optometry. 2017 Mar;94(3):290. DOI: https://doi.org/10.1097/OPX.0000000000001055
- Saraf SS, Ryu CL, Ober MD. The effects of cataract surgery on patients with wet macular degeneration. American journal of ophthalmology. 2015 Sep 1;160(3):487-92. DOI: https://doi.org/10.1016/j.ajo.2015.06.006
- Kessel L, Erngaard D, Flesner P, Andresen J, Tendal B, Hjortdal J. Cataract surgery and age‐related macular degeneration. An evidence‐based update. Acta ophthalmologica. 2015 Nov;93(7):593-600. DOI: https://doi.org/10.1111/aos.12665
- Vicente-Tejedor J, Marchena M, Ramírez L, García-Ayuso D, Gómez-Vicente V, Sánchez-Ramos C, de la Villa P, Germain F. Removal of the blue component of light significantly decreases retinal damage after high intensity exposure. PloS one. 2018 Mar 15;13(3):e0194218. DOI: https://doi.org/10.1371/journal.pone.0194218
- Ratnayake K, Payton JL, Lakmal OH, Karunarathne A. Blue light excited retinal intercepts cellular signaling. Scientific reports. 2018 Jul 5;8(1):1-6. DOI: https://doi.org/10.1038/s41598-018-28254-8
- Peeters A, Magliano DJ, Stevens J, Duncan BB, Klein R, Wong TY. Changes in abdominal obesity and age-related macular degeneration: the Atherosclerosis Risk in Communities Study. Archives of ophthalmology. 2008 Nov 10;126(11):1554-60. DOI: https://doi.org/10.1001/archopht.126.11.1554
- Zhang QY, Tie LJ, Wu SS, Lv PL, Huang HW, Wang WQ, Wang H, Ma L. Overweight, obesity, and risk of age-related macular degeneration. Investigative ophthalmology & visual science. 2016 Mar 1;57(3):1276-83. DOI: https://doi.org/10.1167/iovs.15-18637
- Gopinath B, Flood VM, Louie JC, Wang JJ, Burlutsky G, Rochtchina E, Mitchell P. Consumption of dairy products and the 15-year incidence of age-related macular degeneration. British journal of nutrition. 2014 May;111(9):1673-9. DOI: https://doi.org/10.1017/S000711451300408X
- Kakigi CL, Singh K, Wang SY, Enanoria WT, Lin SC. Self-reported calcium supplementation and age-related macular degeneration. JAMA ophthalmology. 2015 Jul 1;133(7):746-54. DOI: https://doi.org/10.1001/jamaophthalmol.2015.0514
- Umeda S, Ayyagari R, Allikmets R, Suzuki MT, Karoukis AJ, Ambasudhan R, Zernant J, Okamoto H, Ono F, Terao K, Mizota A. Early-onset macular degeneration with drusen in a cynomolgus monkey (Macaca fascicularis) pedigree: exclusion of 13 candidate genes and loci. Investigative ophthalmology & visual science. 2005 Feb 1;46(2):683-91. DOI: https://doi.org/10.1167/iovs.04-1031
- Dawson WW, Dawson JC, Lake KP, Gonzalez-Martinez J. Maculas, monkeys, models, AMD and aging. Vision research. 2008 Feb 1;48(3):360-5. DOI: https://doi.org/10.1016/j.visres.2007.08.006
- Fletcher EL, Jobling AI, Greferath U, Mills SA, Waugh M, Ho T, de Iongh RU, Phipps JA, Vessey KA. Studying age-related macular degeneration using animal models. Optometry and Vision Science. 2014 Aug;91(8):878. DOI: https://doi.org/10.1097/OPX.0000000000000322
- Li H, Chintalapudi SR, Jablonski MM. Current drug and molecular therapies for the treatment of atrophic age-related macular degeneration: phase I to phase III clinical development. Expert opinion on investigational drugs. 2017 Oct 3;26(10):1103-14. DOI: https://doi.org/10.1080/13543784.2017.1369042
- Ellis S, Buchberger A, Holder J, Hughes J. GT005, a gene therapy for the treatment of dry age-related macular degeneration (AMD). Investigative Ophthalmology & Visual Science. 2020 Jun 10;61(7):2295-.
- Schwartz SD, Hubschman JP, Heilwell G, Franco-Cardenas V, Pan CK, Ostrick RM, Mickunas E, Gay R, Klimanskaya I, Lanza R. Embryonic stem cell trials for macular degeneration: a preliminary report. The Lancet. 2012 Feb 25;379(9817):713-20. DOI: https://doi.org/10.1016/S0140-6736(12)60028-2
- Sergejeva O, Botov R, Liutkevičienė R, Kriaučiūnienė L. Genetic factors associated with the development of age-related macular degeneration. Medicina. 2016 Apr;52(2):79-88. DOI: https://doi.org/10.1016/j.medici.2016.02.004
- Francis PJ, Schultz DW, Hamon S, Ott J, Weleber RG, Klein ML. Haplotypes in the complement factor H (CFH) gene: associations with drusen and advanced age-related macular degeneration. PloS one. 2007 Nov 28;2(11):e1197. DOI: https://doi.org/10.1371/journal.pone.0001197
- Landowski M, Kelly U, Klingeborn M, Groelle M, Ding JD, Grigsby D, Rickman CB. Human complement factor H Y402H polymorphism causes an age-related macular degeneration phenotype and lipoprotein dysregulation in mice. Proceedings of the National Academy of Sciences. 2019 Feb 26;116(9):3703-11. DOI: https://doi.org/10.1073/pnas.1814014116
- Clark SJ, Bishop PN, Day AJ. Complement factor H and age-related macular degeneration: the role of glycosaminoglycan recognition in disease pathology. Biochemical Society Transactions. 2010 Oct 1;38(5):1342-8. DOI: https://doi.org/10.1042/BST0381342
- Grassmann F, Harsch S, Brandl C, Kiel C, Nürnberg P, Toliat MR, Fleckenstein M, Pfau M, Schmitz-Valckenberg S, Holz FG, Chew EY. Assessment of novel genome-wide significant gene loci and lesion growth in geographic atrophy secondary to age-related macular degeneration. JAMA ophthalmology. 2019 Aug 1;137(8):867-76. DOI: https://doi.org/10.1001/jamaophthalmol.2019.1318.
- Park SH, Su R, Jeong J, Guo SZ, Qiu K, Joung D, Meng F, McAlpine MC. 3D printed polymer photodetectors. Advanced Materials. 2018 Oct;30(40):1803980. DOI: https://doi.org/10.1002/adma.201803980
- Kotecha A, Zhong J, Stewart D, da Cruz L. The Argus II prosthesis facilitates reaching and grasping tasks: a case series. BMC ophthalmology. 2014 Dec;14(1):1-8. DOI: https://doi.org/10.1186/1471-2415-14-71.
- Sharma R, Khristov V, Rising A, Jha BS, Dejene R, Hotaling N, Li Y, Stoddard J, Stankewicz C, Wan Q, Zhang C. Clinical-grade stem cell–derived retinal pigment epithelium patch rescues retinal degeneration in rodents and pigs. Science translational medicine. 2019 Jan 16;11(475). DOI: https://doi.org/10.1126/scitranslmed.aat5580
- Liu Y, Xu HW, Wang L, Li SY, Zhao CJ, Hao J, Li QY, Zhao TT, Wu W, Wang Y, Zhou Q. Human embryonic stem cell-derived retinal pigment epithelium transplants as a potential treatment for wet age-related macular degeneration. Cell discovery. 2018 Sep 11;4(1):1-0. DOI: https://doi.org/10.1038/s41421-018-0053-y
- Singh MS, MacLaren RE. Stem cell treatment for age-related macular degeneration: the challenges. Investigative ophthalmology & visual science. 2018 Mar 20;59(4):AMD78-82. DOI: https://doi.org/10.1167/iovs.18-24426
- da Cruz L, Fynes K, Georgiadis O, Kerby J, Luo YH, Ahmado A, Vernon A, Daniels JT, Nommiste B, Hasan SM, Gooljar SB. Phase 1 clinical study of an embryonic stem cell–derived retinal pigment epithelium patch in age-related macular degeneration. Nature biotechnology. 2018 Apr;36(4):328. DOI: https://doi.org/10.1038/nbt.4114.
- De Jong PT. Elusive drusen and changing terminology of AMD. Eye. 2018 May;32(5):904-14. DOI: https://doi.org/10.1038/eye.2017.298.
- West SK. Looking forward to 20/20: a focus on the epidemiology of eye diseases. Epidemiologic reviews. 2000 Jan 1;22(1):64-70. DOI: https://doi.org/10.1093/oxfordjournals.epirev.a018025
- Wong WL, Su X, Li X, Cheung CM, Klein R, Cheng CY, Wong TY. Global prevalence of age-related macular degeneration and disease burden projection for 2020 and 2040: a systematic review and meta-analysis. The Lancet Global Health. 2014 Feb 1;2(2):e106-16. DOI: https://doi.org/10.1016/S2214-109X(13)70145-1
- Lindekleiv H, Erke MG. Projected prevalence of age‐related macular degeneration in Scandinavia 2012–2040. Acta ophthalmologica. 2013 Jun;91(4):307-11. DOI: https://doi.org/10.1111/j.1755-3768.2012.02399.x
- Cruickshanks KJ, Nondahl DM, Johnson LJ, Dalton DS, Fisher ME, Huang GH, Klein BE, Klein R, Schubert CR. Generational differences in the 5-year incidence of age-related macular degeneration. JAMA ophthalmology. 2017 Dec 1;135(12):1417-23. DOI: https://doi.org/10.1001/jamaophthalmol.2017.5001
- Colijn JM, Buitendijk GH, Prokofyeva E, Alves D, Cachulo ML, Khawaja AP, Cougnard-Gregoire A, Merle BM, Korb C, Erke MG, Bron A. Prevalence of age-related macular degeneration in Europe: the past and the future. Ophthalmology. 2017 Dec 1;124(12):1753-63. DOI: https://doi.org/10.1016/j.ophtha.2017.05.035
- Samarakoon SM, Chandola HM, Ravishankar B. Effect of dietary, social, and lifestyle determinants of accelerated aging and its common clinical presentation: A survey study. Ayu. 2011 Jul;32(3):315. DOI: https://doi.org/10.4103/0974-8520.93906
- Elliott, R.. “Some Biological Factors in Aging.” (1955).